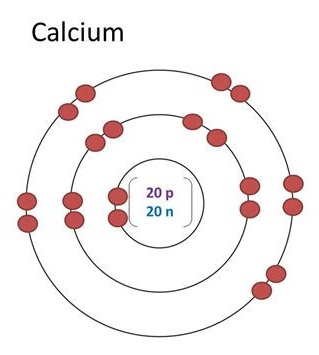 
Up to date, curated data provided by Mathematica 's ElementData function from Wolfram Research, Inc. Also replace URL for the actual url of this page (The stay, ok?). Notes on the properties of Calcium: Specific Heat: Value given for solid phase. Now replace dd, mmmm and yyyy with the day, month, and year you browsed this page. "Atomic Radius of Calcium (Ca) [& State, Uses, Discovery. To make your life (and citation) easier just copy & paste the information below into your assignment or essay: That gives credibility to your paper and it is sometimes required in higher education. properties because a magnesium atom and a calcium atom have the same. When you need to include a fact or piece of information in an assignment or essay you should also include where and how you found that piece of information. How about an incentive to share this post? (You will help other colleagues find this blog)ĭownload and enjoy this complete and colored periodic table for you to edit and enjoy. Need an editable periodic table to edit? Maybe add your school logo, work team or anything else to make your paper look cool?Īlong with basic atom / element information (like Calcium atomic radius and all the other atomic data), it also comes with color coded info about: State (Gas, Liquid or Solid at room temperature), Groups/series details and much more. How a small number of atoms can be joined and form completely different substances.Note 2 pc: 1 atom/unit cell boc: 2 atoms/unit cell fcc: 4 atoms/unit cell Primitive Cubic (pc): 2 x (atomic radius) edge. One atomic mass unit is equal to 1.66 x 10 -24 grams. Metallic Calcium crystallized in a face-centered cubic (fcc) lattice and the atomic radius of calcium is 1.97 A. The unit of measure for mass is the atomic mass unit (amu). Assuming that calcium has an atomic radius of 197 pm, calculate the density of solid calcium. Calculate a value for the atomic radius of calcium. Calcium, Strontium Strontium, Barium Barium, Radium. 
Atomic radius pm Question: Calcium has a face-centered cubic unit cell. Calcium (Ca) and magnsium (Mg) are ubiquitous the rest is rarer. Calcium is atomic number 20 with symbol Ca on the periodic table. Calculate a value for the atomic radius of calcium. Overview: Name: Calcium: Symbol: Ca: Atomic Number: 20: Atomic Weight: 40. The concept can be extended to solvated ions in liquid solutions taking into consideration the solvation shell. Typical values range from 31 pm (0.3 Å) to over 200 pm (2 Å). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Find step-by-step Chemistry solutions and your answer to the following textbook question: Calcium has a cubic closest packed structure as a solid. Get facts about the chemical and physical properties of the element calcium. Technical data for Calcium Click any property name to see plots of that property for all the elements. Ionic radii are typically given in units of either picometers (pm) or angstroms (Å), with 1 Å 100 pm. Note that each element may contain more isotopes. Want to learn more details and data about Calcium (Ca)? Check my Elements Comprehensive List.Īre you having trouble understanding the basics of atomic elements? This video will walk you through: Atomic Mass of Calcium Atomic mass of Calcium is 40.078 u. Fifth most abundant element in the earth's crust (41,500 ppm). Pure metal is produced by replacing the calcium in lime (calcium carbonate, CaCO3) with aluminium in hot, low pressure retorts.įairly hard, silvery-white metal. On moving from top to bottom in a group, the atomic radius increases. Obtained from minerals like chalk, limestone & marble. This is because sodium and potassium are elements of group 1. Virtually no use for the pure metal, however two of its compounds are, lime (CaO) and gypsum (CaSO4), are in great demand by a number of industries. The atomic radius is the distance from the nucleus of an atom to the outermost electrons. Used by many forms of life to make shells and bones. In the case of Calcium the atomic radius is 2.23 Å. There are cool facts about Calcium that most don't know about. Note: Learn more about the atomic radius here. Ok, so what is the atomic radius of an atom of Ca? The density of calcium is 1.54 g / c m 3 g/cm^3 g / c m 3.All atoms have a (theoretical) atomic radius, even Calcium. Usually the atomic radius is calculated through study of lattice structure of various molecules. In order to calculate the density (d) of the metal, we need to know the mass (m) and the volume of the unit.įirstly, let's see the calculation of the mass. Cubic closest packed structure means face centered cubic unit cell and that also means 4 atoms per unit cell (for more explanation see 12.87 c) excercise solved.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
